Projet ANR HAPAR 2014-2019
HAPAR: The paradox of being a specialist for a parasite of marine blooming dinoflagellates
Coordinator : Laure Guillou
Harmful Algal Blooms (HAB) result from the rapid increase and accumulation of opportunistic and often toxic microalgae, most of them belonging to the dinoflagellate group.
Anthropogenic nutrient enrichment (a.k.a. cultural eutrophication) has been pointed out as one of the main factors enhancing HAB frequency, intensity and geographical extension, as observed over the past decades (Anderson et al. 2002).
While a lot of studies have aimed at understanding and predicting the conditions leading to the induction and development of toxic algal blooms, almost nothing is known about the resilience of affected marine coastal ecosystems over time and the role of top down controls over HAB phenomena.
Parasitism is a frequent lifestyle in nature and a major source of evolutionary pressure for both the hosts and parasites.
Given the ubiquity of host-parasite interactions, understanding the factors that generate, maintain, and constrain these associations is of primary interest with implications for a wide range of ecological issues, including dynamics of emerging infectious diseases and invasions (Daszak et al. 2000, Keane and Crawley 2002).
As for invasive plants and animals, the recent increase of the frequency of massive inshore dinoflagellate blooms may well originate from geographical and temporary disruptions between microalgae and their natural enemies (Chambouvet et al. 2008). A
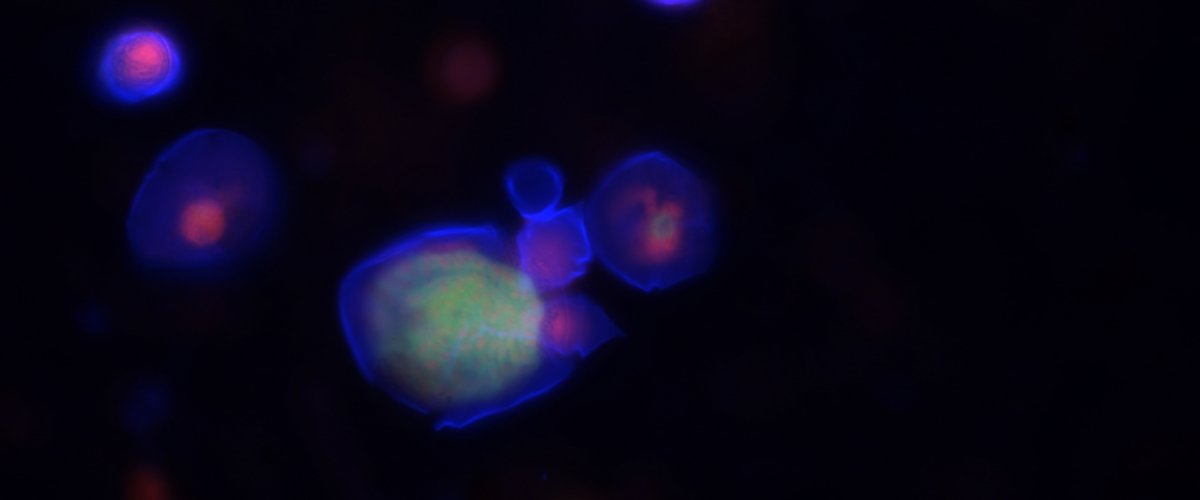
lthough little studied, many extremely virulent microeukaryotic parasites infecting microalgae have been detected in the marine plankton. Syndiniales (also called Marine Alveolates or MALV), which constitute a diverse and highly widespread group (Guillou et al. 2008), are parasitoids (i.e. obligatorily killing their host to complete their life cycle).
Because of their virulence and abundant offspring, such parasites have the potential to control dinoflagellate populations, and therefore toxic microalgal blooms (Montagnes et al. 2008). Noteworthy, Syndiniales are not the only group of protistean parasites infecting marine blooming dinoflagellates, as Perkinsozoa and fungi have also been identified as efficient dinoflagellate parasites (Lepelletier et al. 2014a*, b*). However, Syndiniales is the only group known to gather specialised parasites.
In culture, they exhibit a narrow host spectrum, generally infecting no more than 1-2 dinoflagellate species. In the field, the same parasitic species may infect the same host species, years after years (Chambouvet et al. 2008).
Such specialization strongly relies on the life-history traits and ecology of the host. In contrast to the well-known parasitic relationships encountered in the terrestrial environment, we are far from fully understanding the processes involved in microalga-parasite interactions in marine systems.
Coastal planktonic ecosystems are by nature characterized by strong environmental fluctuations. First, considering the short generation time and the high dispersal capacity of microalgae, their parasites must adapt at a significantly faster rate than for larger hosts (e.g. plants or animals).
Additionally, microbial communities are characterized by rapid turnovers, with successions of different dominant species over weekly timescales. All of these considerations should theoretically lead to the natural selection of generalistic parasites at the expense of specialists.
Thus, the persistence and ecological success of specialists among marine planktonic parasites is an intriguing paradox and a potential limitation to dinoflagellate blooms.
Our overarching goal is to unveil the molecular components, mechanisms and evolutionary forces that determine the ability of specialized parasites to infect their primary host and adapt to a novel host, and how frequent host-range variations could be in nature.